Using natural enemies to control pests is a green, safe and effective way of biological control. Parasitic insects, especially parasitic wasps, are a class of Hymenoptera parasites that can parasitize other insects and arthropods. It is an important natural enemy of agroforestry pests and is widely used in the sustainable control of insect pests. Parasitic insects, unlike predatory insects, usually do not kill the prey directly. While, through regulating the host's physiology and development, they are able to ensure that their offspring can survive and kill the host. Previous studies have shown that parasitic wasps have some specific factors, such as venom, multiple DNA viruses (a class of parasitic wasps’ specific symbiotic virus, polydnavirus (PDV), whose genome is embedded into the genome of the parasitic wasp) and the malformed cells (teratocyte, a special type of cells isolated from embryo serous of parasitic bee, which is distributed in the host blood cavity). These factors may regulate the growth, immune, nutrient composition and metabolism, reproductive system, endocrine system, and ensure the growth and development of their offspring inside or on the host. While so far, its molecular mechanism is still unclear.
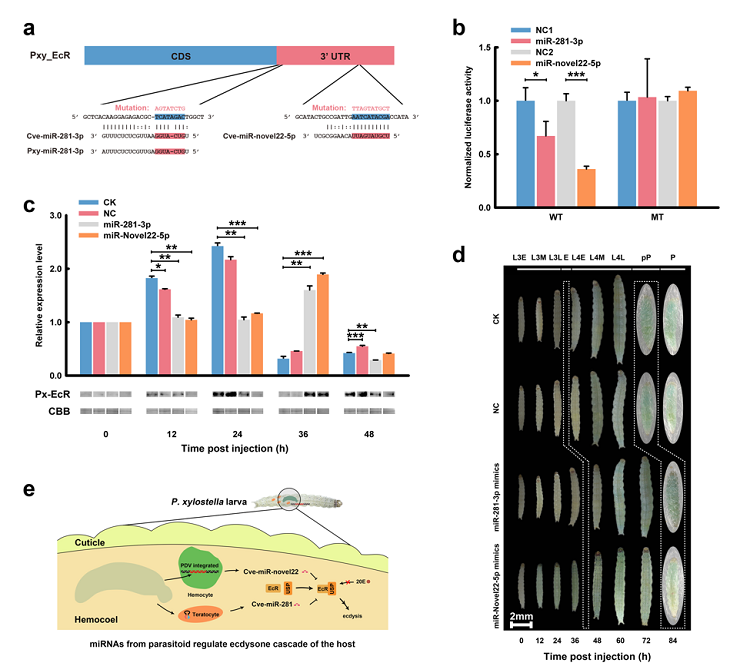
After years of research, Professor Chen Xuexin found that carry factors of parasitic wasps can carry or secrete miRNA into the host and regulate the development of the host. Based on the study of the international important vegetable pest, Plutella xylostella and its natural enemy, Cotesia vestalis, the malformed cells of the Plutella xylostella are found to produce a large number of specific miRNA. Further studies have found that the highly expressed miRNA in the deformed cells can be secreted and absorbed by the blood cells of the host. The miRNA from malformed cells have homologous structures inside host insects, while the increased miRNA in host blood cells mainly comes from miRNA secreted by malformed cells, with a few of them caused by the up regulation of the host's own homologous miRNA. At the same time, the analysis of the miRNA precursor of malformed cells found that 20 of the miRNA were distributed in the parasitic wasp genome in the region of “DNA virus (CvBV) pre virus region”, and could produce specific miRNA after entering the host. Interestingly, unlike the miRNA encoded by the malformed cells, the CvBV encoded miRNA had no homologous in the host. The expression of miRNA in the host blood cells increased by CvBV after its expression. Subsequently, the research team predicted the target gene for these miRNA, and found that the target genes involved in many physiological systems, such as the immune system, nervous system, protein phosphorylation, cell apoptosis and so on. The research team then selected three miRNA with the same target gene. Through functional test (Figure 1), it was found that the miRNA of the malformed cell source and the miRNA of CvBV source can all act on the ecdysone receptor (ecdysone receptor, ECR) of the host Plutella xylostella (ecdysone receptor, ECR), and down regulate the expression of the host ECR and its protein level, and delay the molt and pupationof the diamondback moth, which is consistent with the phenotypes of delayed development. It is the first report that parasitic wasps can produce a variety of miRNA through malformed cells and PDV, and these miRNA can enter the host through different ways, which can effectively regulate the host development. This is also the first report that parasitic animals can regulate the growth and development of host by secreting miRNA.
The first authors of the paper are Zhizhi Wang, who is an assistant researcher; Dr. Xiqian Ye, a graduate student; associate professor Min Shi; Professor Fei Li, and Dr. Zehua Wang, a graduate student. The corresponding author is Professor Xuexin Chen. The other coauthors include Dr Xiaodong Fang from BGI, Professor Shujun Wei from Beijing Academy of agricultural and Forestry Sciences, and Professor Michael R. Strand from University of Georgia. The research has been supported by the National Natural Science Foundation's key projects (31630060), the national key basic research development plan (973 Plan, 2013CB127600) and the innovative group project (31321063).
The link of the paper: https://www.nature.com/articles/s41467-018-04504-1